Products
PROPICONAZOLE
Fungicide
FRAC 3; DMI: triazole
NOMENCLATURE
Common name propiconazole (BSI, draft E-ISO, (m) draft F-ISO)
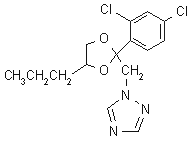
IUPAC name (-1-[2-(2,4-dichlorophenyl)-4-propyl-1,3-dioxolan-2-ylmethyl]-1H-1,2,4-triazole
Chemical Abstracts name 1-[[2-(2,4-dichlorophenyl)-4-propyl-1,3-dioxolan-2-yl]methyl]-1H-1,2,4-triazole
CAS RN [60207-90-1] unstated stereochemistry EEC
no. 262-104-4 Development codes CGA 64 250 (Ciba-Geigy)
PHYSICAL CHEMISTRY
Mol. wt. 342.2 M.f. C15H17Cl2N3O2 Form Yellowish,
odourless, viscous liquid (tech.). B.p. 120 °C (1.9
Pa); >250 °C (101 kPa) V.p. 2.7 × 10-2 mPa (20 °C);
5.6 × 10-2 mPa (25 ºC) KOW logP = 3.72 (pH 6.6, 25 ºC) Henry 9.2 × 10-5
Pa m3 mol-1 (20 °C, calc.) S.g./density 1.29 (20 ºC) Solubility In
water 100 mg/l (20 ºC). In n-hexane 47 g/l. Completely miscible with ethanol,
acetone, toluene and n-octanol (25 °C). Stability Stable
up to 320 ºC; no significant hydrolysis. pKa 1.09,
v. weak base
APPLICATIONS
Biochemistry Steroid demethylation (ergosterol biosynthesis)
inhibitor. Mode of action Systemic foliar fungicide with
protective and curative action, with translocation acropetally in the xylem. Uses Systemic
foliar fungicide with a broad range of activity, at 100-150 g/ha. On cereals,
it controls diseases caused by Cochliobolus sativus, Erysiphe graminis, Leptosphaeria
nodorum, Puccinia spp., Pyrenophora teres, Pyrenophora tritici-repentis, Rhynchosporium
secalis, and Septoria spp. In bananas, control of Mycosphaerella musicola and
Mycosphaerella fijiensis var. difformis. Other uses are in turf against Sclerotinia
homoeocarpa, Rhizoctonia solani, Puccinia spp. and Erysiphe graminis; in rice
against Rhizoctonia solani, Helminthosporium oryzae, and dirty panicle complex;
in coffee against Hemileia vastatrix; in peanuts against Cercospora spp.; in
stone fruit against Monilinia spp., Podosphaera spp., Sphaerotheca spp. and Tranzschelia
spp.; in maize against Helminthosporium spp. Formulation types EC;
SC; Emulsifiable gel.
