Products
ETHEPHON
Plant growth regulator: thylene generator
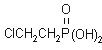
NOMENCLATURE
Common name ethephon (ANSI, Canada); chorethephon
(New Zealand)
IUPAC name 2-chloroethylphosphonic acid
Chemical Abstracts name (2-chloroethyl)phosphonic
acid
CAS RN [16672-87-0] EEC no. 240-718-3
PHYSICAL CHEMISTRY
Mol. wt. 144.5 M.f. C2H6ClO3P Form Colourless
solid; (tech. is a clear liquid). M.p. 74-75 ºC B.p. c. 265 ºC
(decomp.) V.p. <0.01 mPa (20 ºC) KOW logP <-2.20
(25 ºC, unstated pH) Henry <1.55 × 10-9
Pa m3 mol-1 (calc.) S.g./density 1.409.02 (20 ºC)
(tech.) Solubility In water c. 1 kg/l
(23 ºC). Readily soluble in methanol, ethanol, isopropanol, acetone,
diethyl ether, and other polar organic solvents. Sparingly soluble
in non-polar organic solvents such as benzene and toluene. Insoluble
in kerosene and diesel oil. Stability Stable
in aqueous solutions having pH <5. At higher pH, decomposition occurs
with the liberation of ethylene. Sensitive to u.v. irradiation. pKa pKa1
2.5, pKa2 7.2
APPLICATIONS
Mode of action Plant growth regulator with systemic
properties. Penetrates into the plant tissues, and is decomposed to
ethylene, which affects the growth processes. Uses To
promote pre-harvest ripening in apples, currants, blackberries, blueberries,
cranberries, morello cherries, citrus fruit, figs, tomatoes, sugar
beet and fodder beet seed crops, coffee, capsicums, etc.; to accelerate
post-harvest ripening in bananas, mangoes, and citrus fruit; to facilitate
harvesting by loosening of the fruit in currants, gooseberries, cherries,
and apples; to increase flower bud development in young apple trees;
to prevent lodging in cereals, maize, and flax; to induce flowering
of Bromeliads; to stimulate lateral branching in azaleas, geraniums,
and roses; to shorten the stem length in forced daffodils; to induce
flowering and regulate ripening in pineapples; to accelerate boll opening
in cotton; to modify sex expression in cucumbers and squash; to increase
fruit setting and yield in cucumbers; to improve the sturdiness of
onion seed crops; to hasten the yellowing of mature tobacco leaves;
to stimulate latex flow in rubber trees, and resin flow in pine trees;
to stimulate early uniform hull split in walnuts; etc. Formulation
types EC; SL. Compatibility Incompatible
with alkaline materials and with solutions containing metal ions, e.g.
iron-, zinc-, copper-, and manganese-containing fungicides.
