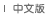About enzymes
Basic Theory Behind Enzymes
Enzyme ¨C biological catalyst . The term enzyme comes from zymosis, the Greek word for fermentation , a process accomplished by yeast cells and long known to the brewing industry, which occupied the attention of many 19th-century chemists.Louis Pasteur recognized in 1860 that enzymes were essential to fermentation but assumed that their catalytic action was inextricably linked with the structure and life of the yeast cell. Not until 1897 was it shown by German chemist Edward B¨ąchner that cell-free extracts of yeast could ferment sugars to alcohol and carbon dioxide; B¨ąchner denoted his preparation zymase. This important achievement was the first indication that enzymes could function independently of the cell.
The first enzyme molecule to be isolated in pure crystalline form was urease, prepared from the jack bean in 1926 by American biochemist J. B. Sumner, who suggested, contrary to prevailing opinion, that the molecule was a protein . In the period from 1930 to 1936, pepsin , chymotrypsin , and trypsin were successfully crystallized; it was confirmed that the crystals were protein, and the protein nature of enzymes was thereby firmly established.
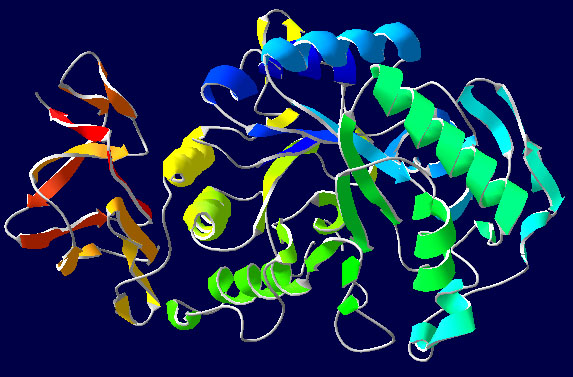
Figure 1: 3D Structural Diagram of an ¦Á-Amylase
Catalysis by an enzyme is analogous to removing a pebble that is stopping the ball from rolling down the hill; the reaction goes to completion more quickly, but the final product is identical. An enzyme contains one or more binding sites where the substrate(s) attach, and active site(s), where the amino acids perform the catalysis; and frequently one or more other binding sites that serve regulatory functions, which increase or inhibit the enzyme's activity. Enzymes are usually specific as to the reactions they catalyze and the substrates that are involved in these reactions. Complementary structural properties of the enzyme and substrate are responsible for this specificity.
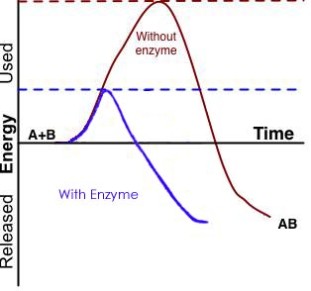 Figure 2: Diagram of a catalytic reaction, showing the energy needed at each stage of the reaction. The substrates (A and B) normally need a large amount of energy to reach the transition state, which then reacts to form the end product (AB). The enzyme forms a microenvironment in which A and B can reach the transition state more easily, reducing the amount of energy required. Since the lower energy state is easier to reach and therefore occurs more frequently, as a result the reaction is more likely to take place, thus improving the reaction speed.
Figure 2: Diagram of a catalytic reaction, showing the energy needed at each stage of the reaction. The substrates (A and B) normally need a large amount of energy to reach the transition state, which then reacts to form the end product (AB). The enzyme forms a microenvironment in which A and B can reach the transition state more easily, reducing the amount of energy required. Since the lower energy state is easier to reach and therefore occurs more frequently, as a result the reaction is more likely to take place, thus improving the reaction speed. Most enzymatic reactions occur within a relatively narrow temperature range (usually from about 30ˇăC to 40ˇăC), a feature that reflects their complexity as biological molecules. Each enzyme has an optimal range of p H for activity; for example, pepsin in the stomach has maximal reactivity under the extremely acid conditions of p H 1-3. Effective catalysis also depends crucially upon maintenance of the molecule's elaborate three-dimensional structure.
Loss of structural integrity, which may result from such factors as changes in pH or high temperatures, almost always leads to a loss of enzymatic activity. An enzyme that has been so altered is said to be denatured, meaning it as lost its native, higher-order structure of protein molecules in solution. Most globular proteins exhibit complicated three-dimensional folding described as secondary, tertiary, and quarternary structures.

Figure 3: Bacteria Culture dedicated to the production of ZDSS's Acidic Protease
These conformations of the protein molecule are rather fragile, and any factor that alters the precise geometry is said to cause denaturation. Thus, the denatured enzyme is often without catalytic function. Renaturation is accomplished with varying success, and occasionally with a return of biological function, by exposing the denatured protein to a solution that approximates normal physiological conditions.
These conformations of the protein molecule are rather fragile, and any factor that alters the precise geometry is said to cause denaturation. Thus, the denatured enzyme is often without catalytic function. Renaturation is accomplished with varying success, and occasionally with a return of biological function, by exposing the denatured protein to a solution that approximates normal physiological conditions.
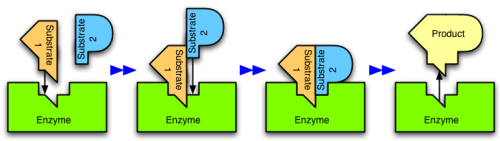
Figure 4: An enzyme catalyzes the reaction of two substrates to form one product.
Loss of structural integrity, which may result from such factors as changes in pH or high temperatures, almost always leads to a loss of enzymatic activity. An enzyme that has been so altered is said to be denatured, meaning it as lost its native, higher-order structure of protein molecules in solution. Most globular proteins exhibit complicated three-dimensional folding described as secondary, tertiary, and quarternary structures.

Figure 3: Bacteria Culture dedicated to the production of ZDSS's Acidic Protease
These conformations of the protein molecule are rather fragile, and any factor that alters the precise geometry is said to cause denaturation. Thus, the denatured enzyme is often without catalytic function. Renaturation is accomplished with varying success, and occasionally with a return of biological function, by exposing the denatured protein to a solution that approximates normal physiological conditions.

Figure 4: An enzyme catalyzes the reaction of two substrates to form one product.